CAR-NK Cells: Breaking Barriers in Solid Tumor Immunotherapy
Over the past decade, chimeric antigen receptor (CAR) T‑cell therapy has revolutionized the treatment of hematological malignancies. However, translating this success to solid tumors remains challenging due to insufficient infiltration, the immunosuppressive tumor microenvironment (TME), and antigen heterogeneity. Natural killer (NK) cells-first described in 1975-offer unique advantages: they exert spontaneous cytotoxicity without prior sensitization, are not restricted by major histocompatibility complex (MHC), and can be sourced from multiple allogeneic reservoirs. CAR‑NK cell therapy combines the precision of CAR targeting with the innate cytotoxic machinery of NK cells, emerging as a promising alternative or complement to CAR‑T cells for solid tumors.
1. Diverse Cell Sources for CAR NK Generation
NK cells are innate lymphocytes that recognize stressed or malignant cells through a balanced array of activating and inhibitory receptors. Their MHC‑independent recognition allows safe allogeneic use. The clinical translation of CAR‑NK relies on scalable, well‑characterized cell sources, each with distinct advantages and limitations.
Table 1. Comparison of NK Cell Sources for CAR‑NK Engineering
| NK Cell Sources | Advantages | Limitations |
|---|---|---|
| Cord blood (CB) | Global banking availability; high proliferative potential | Ex vivo expansion required (≈2 weeks); donor variability |
| iPSC‑derived | Unlimited expansion; homogeneous, standardized product | Immature phenotype; need genetic modification for ADCC; prolonged culture |
| Peripheral blood (PB) | Mature, highly functional and cytotoxic | Ex vivo expansion; donor variability and availability |
| NK‑92 cell line | High proliferation; easily engineered; uniform product | Irradiation required → limited persistence; low/absent CD16 → weak ADCC |
| Cord blood HSPCs | Readily available NK precursors | Long expansion (5‑6 weeks); low CD16 expression; donor variability |
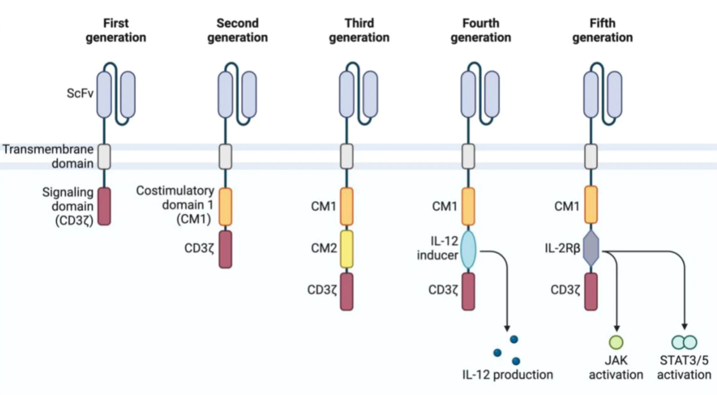
2. CAR Construction: From First to Fifth Generation
Chimeric antigen receptors consist of an extracellular antigen‑binding domain (usually a single‑chain variable fragment, scFv), a hinge and transmembrane region, and intracellular signaling domains. The evolution of CAR design has been driven by the need to enhance persistence, activation, and safety. For CAR‑NK cells, NK‑specific signaling domains (2B4, DNAM‑1, NKG2D) and adapter moieties (DAP10/DAP12) are increasingly used to optimize activation and cytotoxicity.
3. Strategies to Overcome Solid Tumor Microenvironment Barriers
The solid TME imposes multiple layers of resistance: antigen heterogeneity, physical barriers (stroma, extracellular matrix), hypoxia, immunosuppressive metabolites, and checkpoint ligands. Advanced CAR‑NK engineering now addresses each obstacle.
3.1 Counteracting Antigen Heterogeneity & Immune Escape
- High‑affinity scFvs: Generated via phage display or computational design to recognize low‑density tumor‑associated antigens (TAAs).
- Multi‑target CARs: Dual‑specific CAR‑NK cells (e.g., PD‑L1/HER2, GD2/NKG2D ligands) reduce antigen escape.
- BiKEs/TriKEs: Bispecific or trispecific engagers bridge NK receptors with tumor antigens, boosting activation and persistence (TriKEs often incorporate IL‑15).
- NK‑optimized signaling domains: 2B4, DAP10/DAP12 enhance activation in the TME.
3.2 Preventing Trogocytosis and Fratricide
- Dual AI‑CAR system: Co‑expression of an activating CAR (aCAR) against tumor antigen and an inhibitory CAR (iCAR) recognizing an NK‑specific antigen (e.g., CS1) prevents self‑killing after antigen acquisition.
- Pharmacological inhibition: Pre‑treatment with synaptic inhibitors (latrunculin A, cytochalasin D) blocks trogocytosis.
3.3 Overcoming Physical Barriers (Stroma & ECM)
- Targeting cancer‑associated fibroblasts (CAFs): FAP‑CAR NK cells or anti‑fibrotic drugs (nintedanib) reduce stromal obstruction.
- ECM degradation: Engineering CAR‑NK to secrete ECM‑remodeling enzymes (heparanase, MMPs, collagenase).
- Regional delivery: Intraperitoneal or hepatic artery infusion bypasses physical barriers and increases local concentration.
3.4 Enhancing Chemotaxis and Infiltration
- Chemokine receptor engineering: Forced expression of CXCR1/2/4, CCR4/5/7 matching tumor chemokine profiles. For instance, CXCR2‑modified mesothelin‑CAR‑NK significantly improves pancreatic cancer infiltration.
- Cytokine priming: IL‑2/IL‑15/IL‑18 upregulate chemokine receptors such as CCR7.
- Combination with oncolytic viruses or radiotherapy: Modulate TME to enhance lymphocyte recruitment.
3.5 Tackling Hypoxia and Metabolic Stress
- HIF‑1α blockade: Restores expression of activating receptors (NKG2D, NKp46) and cytotoxicity.
- Metabolic reprogramming: Blocking glycolysis/LDH, activating AMPK/PGC‑1α pathways improves mitochondrial fitness.
- Hypoxia‑responsive CAR: CAR expression driven by hypoxia response elements limits on‑target/off‑tumor toxicity.
3.6 Disrupting Adenosine and TGF‑β Suppression
- Adenosine axis: CD73 antibody or CD73‑CAR reduces adenosine; A2A receptor knockout restores cytotoxicity.
- TGF‑β resistance: Knockout of TGFβR2, expression of dominant‑negative TGFβR, or combination with TGF‑β inhibitors.
- Oxidative stress defense: Co‑expression of catalase clears reactive oxygen species (H2O2) in the TME.
3.7 Improving Persistence and Survival
- Cytokine "armoring": Constitutive or inducible expression of IL‑15 (often combined with IL‑12/IL‑21) provides sustained survival signals.
- Genetic knockout of negative regulators: CISH deletion enhances mTOR signaling and mitochondrial fitness; overexpression of anti‑apoptotic genes (BCL‑2, BCL‑XL) and amino acid transporters (SLC1A5, SLC3A2/SLC7A5) boosts survival.
3.8 Integrating Immune Checkpoint Inhibition
- PD‑1/PD‑L1 axis: PD‑1 signal converters, PD‑1 knockout, PD‑L1‑targeted CAR‑NK combined with IL‑15, or dual CARs (PD‑L1 + HER2).
- Other checkpoints: HLA‑G CAR‑NK with DAP12 signaling; B7‑H3 CAR‑NK retains cytotoxicity even under low pH and hypoxia; CD47/SIRPα blockade via SIRPα‑Fc fusion restores phagocytic and NK function.
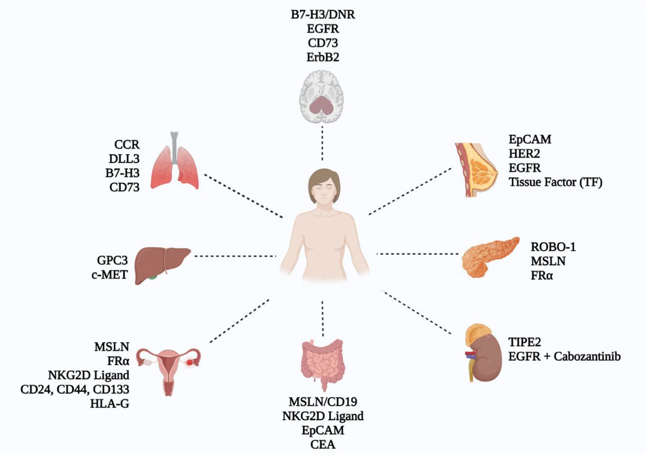
4. Key CAR‑NK Targets in Solid Tumors
Current research has identified a spectrum of tumor‑associated antigens (TAAs) for CAR‑NK therapy across various solid malignancies. These targets are selected based on tumor expression, specificity, and resistance to antigen escape. Many ongoing trials are evaluating multi‑target CAR‑NK constructs to mitigate antigen escape.
5. CAR‑T, CAR‑NK and CAR‑M: A Comparative Overview
While CAR‑T cells have shown remarkable efficacy in hematologic cancers, CAR‑NK cells offer several distinct advantages particularly relevant to solid tumors. The table below summarizes key differences.
| Feature | CAR‑T | CAR‑NK | CAR‑M (Macrophages) |
|---|---|---|---|
| MHC restriction | Yes (HLA‑matching required for allogeneic) | No (MHC‑independent, off‑the‑shelf possible) | No |
| CRS & ICANS risk | High (IL‑6, IFN‑γ driven) | Low (<50% CRS incidence, minimal neurotoxicity) | Moderate |
| GvHD risk | Significant (requires strict HLA matching) | <5% - lacks TCR‑mediated alloreactivity | Low |
| In vivo persistence | Long (months to years) | Short (2‑4 weeks, safer but requires persistence engineering) | Intermediate |
| Killing mechanisms | Perforin/granzyme, FasL | Perforin/granzyme, CD16‑mediated ADCC, TRAIL/FasL, cytokine secretion (IFN‑γ, GM‑CSF) | Phagocytosis, cytokine release |
| Antigen escape vulnerability | High (single antigen loss) | Low - dual CAR + innate receptors (NKG2D, NKp46) recognize stress ligands | Moderate |
| Manufacturing | Autologous or allogeneic (complex) | Allogeneic "off‑the‑shelf" feasible, multiple sources | Allogeneic feasible |
Due to these attributes, CAR‑NK cells are increasingly viewed as a safer, more versatile platform for solid tumor immunotherapy, with dual targeting capacity that counters tumor heterogeneity.
Conclusions
Successful clinical translation of CAR‑NK therapy for solid tumors demands a multi‑pronged strategy to overcome antigen heterogeneity, physical and immunosuppressive barriers, and metabolic stress. Early‑phase trials have validated the feasibility of multi‑antigen targeting, IL‑15 arming, and regional administration. Moving forward, precise combination approaches tailored to specific TME signatures-such as targeting CAFs plus ECM degradation in desmoplastic tumors, or metabolic reprogramming in hypoxic niches-will be crucial. Moreover, integration of artificial intelligence for optimal CAR design, synergistic partners (oncolytic viruses, checkpoint inhibitors), and standardized "off‑the‑shelf" manufacturing will accelerate clinical adoption.
Outsource CAR-T & CAR-NK Services With Confidence
Our GMP CAR-T & CAR-NK CDMO services provide biopharma innovators with end-to-end manufacturing solutions for next-generation cell therapies.
References:
- Balkhi, Sahar, et al. "CAR-NK cell therapy: promise and challenges in solid tumors." Frontiers in Immunology 16 (2025): 1574742.
- Islam, Fahmida, et al. "CAR-NK Engineering to Overcome TME Barriers." Cells 15.1 (2025): 21.
- Li, Sisi, et al. "Precision sniper for solid tumors: CAR-NK cell therapy." Cancer Immunology, Immunotherapy 74.9 (2025): 275.
- Laskowski, Tamara J., Alexander Biederstädt, and Katayoun Rezvani. "Natural killer cells in antitumour adoptive cell immunotherapy." Nature Reviews Cancer 22.10 (2022): 557-575.

Your email address will not be published. Required fields are marked *